The Plateau and the Squeeze
How an invisible electrical shift across a membrane transforms into a physical force capable of pushing blood through the body.
If the heart were only an electrical organ, the action potential would be enough. The voltage would spike, the wave would travel, and the story would end there — a silent, invisible flash of electricity propagating through the chest. But the heart is a mechanical pump. That electrical wave must somehow grab hold of the physical machinery inside the cell and force it to contract.
The bridge between the electrical world and the mechanical world is calcium. The moment of translation happens during the long, flat Phase 2 of the action potential. This is excitation-contraction coupling.
The Mechanism: Calcium-Induced Calcium Release
The Trigger: L-Type Calcium Channels
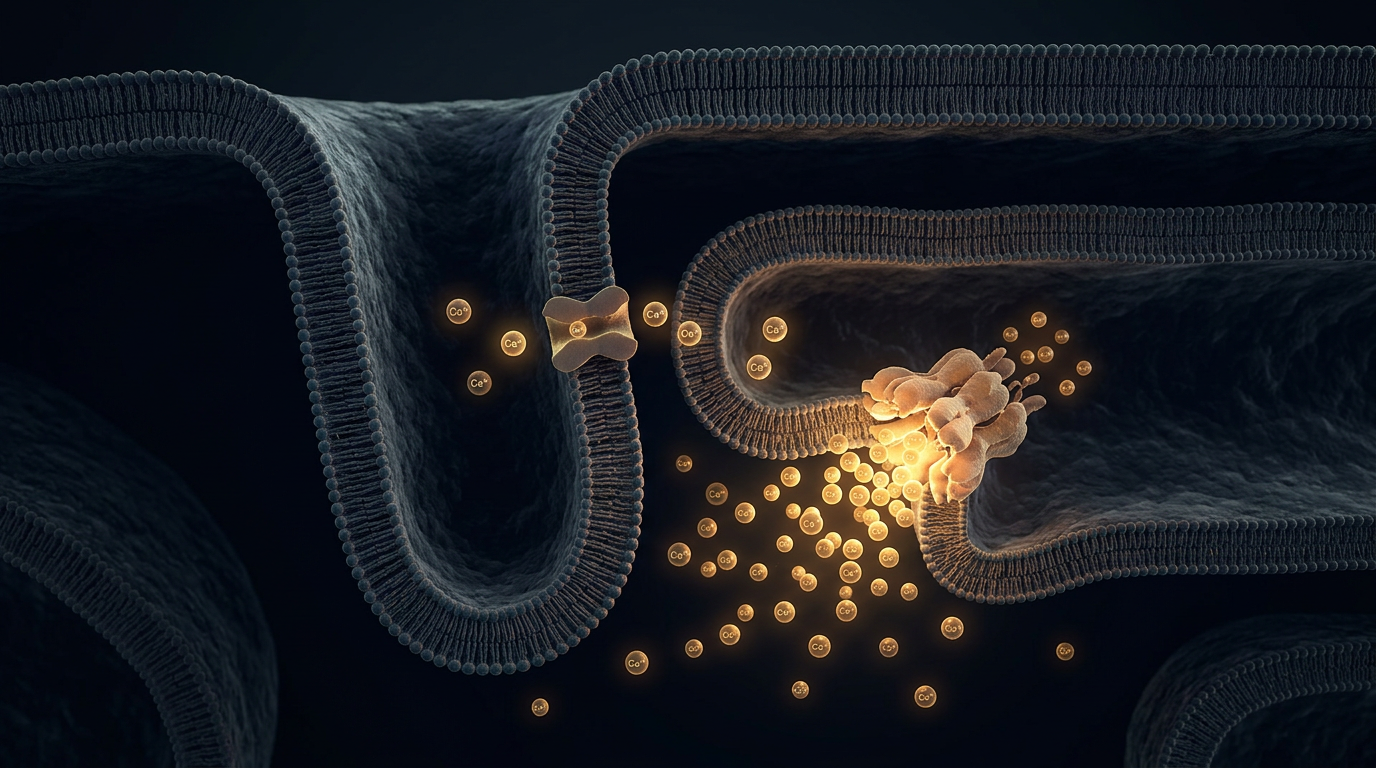
In Chapter 2, we saw that the Phase 2 plateau is maintained by inward calcium current balancing outward potassium current. That calcium enters through L-type calcium channels located in specialized invaginations of the cell membrane called T-tubules. The T-tubules bring the extracellular space deep into the interior of the cell, directly adjacent to the cell's internal calcium warehouse: the sarcoplasmic reticulum (SR).
The amount of calcium that enters through the L-type channels during Phase 2 is small. It is not nearly enough to cause the cell to contract on its own. Instead, it serves as a chemical spark.
The Amplifier: The Ryanodine Receptor
Embedded in the membrane of the sarcoplasmic reticulum are massive, mushroom-shaped proteins called ryanodine receptors (RyR). These are calcium channels, but they don't open in response to voltage. They open when they sense calcium binding to them.
When that small puff of trigger calcium enters through the L-type channel from the outside, it binds to the nearby ryanodine receptors on the SR. The receptors swing open, and a massive flood of calcium pours out of the SR into the cytosol. A tiny extracellular signal has been amplified into a massive intracellular wave. This is exactly what the name implies: Calcium-Induced Calcium Release (CICR).
The Squeeze: Actin and Myosin
The flood of calcium bathes the myofilaments — the structural proteins that actually execute the contraction. Calcium binds to Troponin C, which acts like a physical lock on the actin filament. When calcium binds, Troponin changes shape, rolling a blocking protein (tropomyosin) out of the way.
With the binding sites exposed, myosin heads grab the actin, bend, release, and grab again. The filaments slide past each other, the cell shortens, and the heart muscle contracts. Systole has begun.
Relaxation: Packing the Calcium Away
For the heart to fill with blood again, it must relax (diastole). Relaxation requires removing the calcium from the cytosol so that Troponin C locks up the actin again.
This cleanup is hard work. About 80% of the calcium is actively pumped back into the SR by an ATP-consuming pump called SERCA (Sarcoplasmic Reticulum Ca²⁺-ATPase). The remaining 20% is pushed out of the cell entirely by the Sodium-Calcium Exchanger (NCX), which trades 3 inward sodium ions for 1 outward calcium ion.
The Genius of the Plateau
Why did nature design a 200-millisecond plateau for the cardiac action potential? A skeletal muscle cell fires an action potential that lasts only 2 to 5 milliseconds. It zips through depolarization and repolarization instantly, then twitches.
If you stimulate a skeletal muscle repeatedly and rapidly, the mechanical twitches summate. The muscle locks into a sustained, unyielding contraction called tetanus. You use this when lifting a heavy weight — a smooth, continuous force.
If the heart were to enter tetanus, it would be instantly fatal. A heart locked in systole cannot fill with blood. To prevent this, the heart's electrical refractory period must outlast its mechanical contraction.
That is the true purpose of the Phase 2 plateau. By keeping the voltage depolarized and the sodium channels locked in their inactivated state for hundreds of milliseconds, the cell guarantees it cannot be re-excited until it has fully contracted and begun to relax. The plateau enforces a mandatory diastole. The heart must beat, relax, and fill before it can be told to beat again.
Clinical Takeaway: Heart Failure
In a healthy heart, this system is tightly regulated. When you exercise, sympathetic nerves release norepinephrine, which activates beta-1 receptors. This cascades down to phosphorylate the L-type calcium channels (letting more trigger calcium in) and SERCA (packing calcium away faster). The result is a stronger squeeze and a faster relaxation, perfectly tuned for a high heart rate.
In failing myocardium, this elegant machinery falls apart. The cells are chronically overloaded. SERCA downregulates, meaning the SR struggles to re-pack the calcium during diastole. At the same time, NCX upregulates, desperately trying to pump calcium out of the cell entirely.
The consequence is twofold:
- The SR is depleted of calcium, leading to a weak, pathetic release on the next beat (poor inotropy / systolic dysfunction).
- The reliance on NCX to clear calcium generates a net inward current during diastole (because 3 Na⁺ come in for every 1 Ca²⁺ out). As we learned in Chapter 1, this depolarizing current can trigger a Delayed Afterdepolarization (DAD), placing the failing heart at high risk for lethal ventricular arrhythmias.
Key Takeaways
- Phase 2 is not just an electrical delay; it is the bridge to mechanical contraction via the entry of calcium.
- A small amount of calcium entering through L-type channels triggers a massive release of calcium from the sarcoplasmic reticulum — Calcium-Induced Calcium Release (CICR).
- Relaxation requires active energy to pump calcium back into the SR via SERCA, and out of the cell via NCX.
- The long plateau ensures the electrical refractory period outlasts the mechanical contraction, physically preventing the heart from locking in fatal tetanus.
- Heart failure is fundamentally a breakdown in this calcium handling, leading to both weak contractions and high arrhythmia risk due to DADs.